You must login before you can run this tool.
You are here: resources › Tools › Guidelines Viewer › About
Guidelines Viewer
View Guidelines in Ontology Browser
Version 1.2 - published on 05 Jun 2014
This tool is closed source.
Category
Published on
Abstract
'Guideline Viewer' is a tool for visualizing heuristic knowledge which can't be modeled using mathematical equations. The tool by itself is an ontology editor, called Protege. A package named 'Graph Widget' is used in Protege to visualize the heuristic knowledge in the form of 'Guidelines'. The advantages of the heuristic knowledge stored in the form of 'guidelines' as above is that it is 1) Computer interpretable 2) Portable 3)Sharable and 4) Revisiting the knowledge is possible. The guidelines are built based on ontology called 'Guideline Ontology'. The basis for the ontology is GLIF functionality. GLIF stands for 'GuideLine Interchange Format'. This technology has been widely used by the medical informatics community for sharing the clinical guidelines. The guidelines are built using the basic steps such as state step, branch step, action step, decision step, synchronization step, case step etc. It should be noted that the guideline knowledge is 'STATIC'. The execution of guidelines is possible using the technology called GLEE (GuideLine Execution Engine) developed again by the medical informatics community. It is important to update the knowledge regularly. The 'Guideline Viewer' currently has guidelines built for two aspects 1) 'Formulation aspects such as Route and Excipient Selection' for tablet dosage form and 2) Mill selection for size reduction of API (Active Pharmaceutical Ingredient). The references below are to be used for more information on the guideline tool.
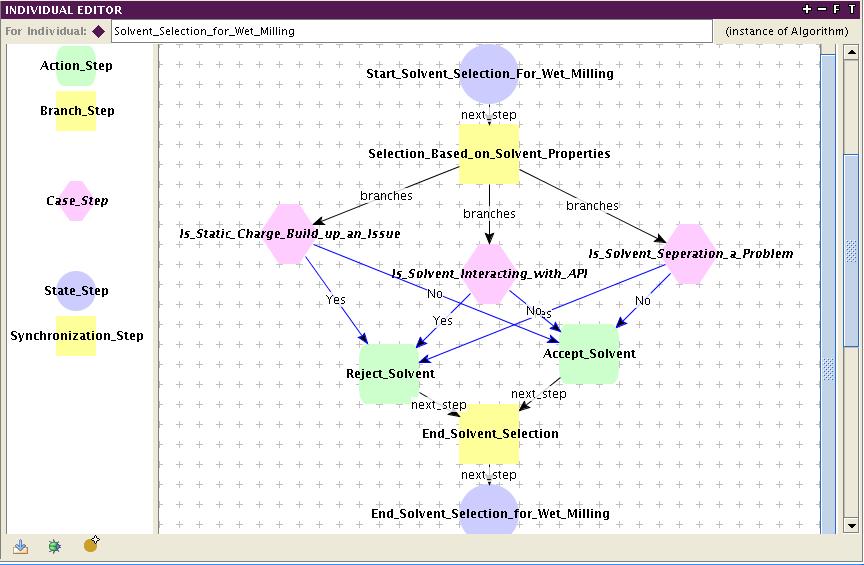
Guideline for solvent selection for wet milling
Credits
The LIPS Laboratory for Intelligent Processing Systems: Venkat Venkatasubramanian, Gintaras Reklaitis, Shuo-Huan Hsu, Chunhua Zhao, Girish Joglekar, Ankur Jain, Leaelaf Hailemariam,Pradeep Suresh and Pavan Akkisetty.
References
1. C. Zhao, A. Jain, L. Hailemariam, P. Suresh, P. Akkisetty, G. Joglekar, V. Venkatasubramanian, G.V. Reklaitis, K. Morris, & P. Basu, 2006, Towards Intelligent Decision Support for Pharmaceutical Product Development, Journal of Pharmaceutical Innovation, Sep-Oct, 23-35
2. V.Venkatasubramanian, C. Zhao, G. Joglekar, A. Jain, L. Hailemariam, P. Suresh,P. Akkisetty, K. Morris, & G.V., Reklaitis, 2006, Ontological Informatics Infrastructure for Pharmaceutical Product Development and Manufacturing, Computers & Chemical Engineering, 30, 10-12, 1482-1496
3. C. Zhao, A., Jain, L. Hailemariam, G. Joglekar, V. Venkatasubramanian, G.V. Reklaitis, & K. Morris, 2006, A Unified Approach for Knowledge Modeling in Pharmaceutical Product Development, in: 16th ESCAPE and 9th International Symposium on Process Systems Engineering, 21B, Marquardt, W., & Pantelides, C. (Eds.), Elsevier, 1929-1935
4. A. Jain, 2007, An Ontological Framework for Knowledge Modeling and Decision Support for Pharmaceutical Product Development, Ph.D. Dissertation, Purdue University, West Lafayette, IN, USA
5. W3C, 2004, Web Ontology Language Overview, W3C Recommendation. Available online at: http://www.w3.org/TR/owl-features/
6. Protégé, url. http://protege.stanford.edu/
7. M. Peleg, A. Boxwala, S. Tu, D. Wang, O. Ogunyemi, and Q. Zeng, (2004), Guideline Interchange Format 3.5 Technical Specification. Available online at: http://www.glif.org
8. Protege-OWL API, url. http://protege.stanford.edu/plugins/owl/api
Cite this work
Researchers should cite this work as follows: